Silica is a chemical that is feared by many reef keepers. Visions of a reef tank covered with diatoms so thick that you can’t see through the glass come to mind. More recently, others have suggested that soluble silica does not, in fact, increase diatom growth in reef tanks. Much of the debate swirls around whether silica sand is a good choice for the substrate in a reef tank. According to different individuals, it can easily release soluble silica, or it cannot possibly do so. In this case it appears that the truth is somewhere between these views.
In this article I will expand on a previous article covering silica in reef tanks by Craig Bingman.1 I will describe the nature of silica in the oceans, and describe which organisms use silica, how they use it, and how much they need. I’ll discuss issues around measurement of soluble and insoluble silica, and also describe some of the sources of soluble silica in reef tanks, including a demonstration that release from “silica” sand can be substantial.
I’ll also show with dosing experiments that soluble silica is rapidly depleted from my reef tank (about 50% per day). When adequate silicate is added to my reef tank, diatom growth appears to increase. Contrary to popular notions, however, the increased diatom growth actually makes my glass easier to see through than the green algae that it replaced.
Finally, and I think most importantly, I’ll recommend that people consider dosing soluble silica to their tanks to support the variety of organisms that potentially use it, from sponges to limpets.
Silica in the Ocean
Dissolved silica in the ocean largely takes the form of silicic acid, Si(OH)4. Since it is acidic and has a pKa somewhat above normal seawater pH values (pKa ~ 9.5 in freshwater; possibly it is a bit lower in seawater), about 5% of it will be present as silicate, Si(OH)3O–. Apparently, many diatoms take up the Si(OH)4 form directly, although there is some evidence that certain organisms take up Si(OH)3O–. In this article, I will not generally refer to silicic acid or silicate unless I am specifying one or the other. Typically I will refer to the sum of them as “soluble silica” or just silica, if the context is clear.
The concentration of soluble silica in the ocean is highly variable. In near surface waters, diatoms are very efficient at sucking it out of solution to make their SiO2 frustules. A diatom bloom in the ocean can drive the concentration of silica down from a value not atypical for the whole ocean, 45 mM (2.7 ppm SiO2), to less than 1 mM (0.06 ppm SiO2), at which point the diatoms can become silicon limited.2 Typical silica concentrations in the surface waters of the equatorial Pacific are a few mM.3
Rivers are the primary input of silica into the oceans (80% of the total input; underwater vents and deposition from the atmosphere are also significant contributors), and river water worldwide averages 150 mM (9 ppm SiO2).4 Consequently, coastal areas near a river may have greater silica concentrations than open ocean areas. This input is approximately balanced by the deposition of silica on the ocean bottom. However, the total biogenic incorporation of silica into organisms is about 40 times as fast as river input, indicating that much of the silicon is deposited into skeletons and re- dissolved many times before it becomes “permanently” incorporated into sea floor sediments. The average residence time for a single silicon atom in the oceans is only about 400 years, before it gets deposited in some fashion.4
When diatoms and radiolarians die and sink, they slowly dissolve, and the silica concentration in deep water can be much higher than surface water for this reason. All ocean waters are undersaturated with respect to amorphous silica (allowing the silica structures in diatoms and radiolarians to dissolve), and most waters are undersaturated even with respect to quartz,5 although its dissolution is kinetically slow, allowing beaches to exist.
Marine Organisms that Use Silica: Diatoms
There are a variety of marine organisms that use silica. In the oceans, the primary consumers are diatoms. They use silica to form frustules that provide them with a hard, silica-containing cell wall. These frustules form a dizzying array of beautiful patterns, and are well represented at the interface between science, art, and photography.6
With one exception (discussed below) all diatoms require silica for growth, and low silica levels cause significant changes in the cell cycle.7 Silicon is a major limiting nutrient for diatom growth in certain parts of the oceans,8 although iron,9 nitrogen, and phosphorus can also be limiting. There have been many studies on the uptake of silica by diatoms. Most diatoms take up silica in the form of silicic acid, although one has been shown to take up the silicate form.10 If absorbing silica is a limiting factor, then it makes sense to transport silicic acid since it is present at much higher concentrations than is silicate, and hence is potentially easier to transport.
Different diatom species have different abilities to absorb silica from the water. That is, as the silica concentration drops, some diatoms can continue to pull silica from the water while others cannot. Most diatoms have half maximal rates of silica absorption of 0.7-10 mM (0.04 – 0.6 ppm SiO2),2 but some are substantially higher, up to about 60 mM (2.6 ppm SiO2).2 The in-situ average for biogenic silica uptake in the surface layer of the equatorial pacific showed half maximal uptake at a silica concentration of 1.6 mM at 3°S and 2.4 mm at the equator, which was close to the silica concentrations present.3
There apparently are genes for many different silica transporters in each of the diatom species that has been investigated.8 Diatoms also somehow maintain internal silicic acid concentrations at levels higher than its solubility, but the mechanism for accomplishing this is unclear. Nevertheless, it is obvious that this facilitates the deposition process, and inhibits dissolution of the existing frustule. Diatoms apparently use proteins to guide the deposition process, where soluble silica is converted into the intricate solid frustule, but exactly how this role is accomplished is not known.8
In a reef tank like mine with silica concentrations below 0.8 mM (0.05 ppm SiO2, the practical limit of the Hach silica kit), some diatoms will have a hard time absorbing silica. Many reef tanks may, in fact, be selecting for diatoms that are able to get enough silica at the low concentrations typically available. Are diatoms silica-limited in reef tanks? That question is addressed experimentally below.
In the oceans, diatoms are silica limited in some natural settings (like the polar regions and the Sargasso Sea, where the ambient silica concentration is less than 1 mM (0.06 ppm SiO2).11 There have also been many cases where eutrophication of natural waters has raised nitrogen and phosphorus levels to the point where silica has become limiting,12 even when it was not limiting in pristine waters. In reef tanks, where nitrogen and phosphorus are often not in short supply, it makes sense that silica could be limiting. In case you were thinking that silica limitation to diatom growth is necessarily a good thing, there are drawbacks. The limitation of silica, inhibiting the growth of diatoms that would otherwise take up the limiting nutrients nitrogen and phosphorus, has even been implicated in blooms of cyanobacteria.1
Marine Organisms that Use Silica: Sponges
Sponges are the second largest consumer of dissolved silica in the ocean, after diatoms.14 Many sponges use silica to form internal structures, called spicules, which help them retain their shape. In the case of the sponge Tethya aurantia, these spicules are needles of amorphous silica that comprise 75% of its dry weight. These spicules are 2 mm long and 30 mm wide.15
Most frequently, the spicules in sponges are hydrated amorphous silica, and also contain collagen, an organic material (although sometimes they are calcium carbonate). In order to direct the formation of these silica spicules, sponges produce enzymes that help control the silica deposition process. In the case of the sponges Suberites domuncula and Tethya auranita, an enzyme called silicatein is produced. When the silicate concentration is increased in the surrounding water from 1 mM (0.06 ppm SiO2) to 60 mM (3.6 ppm SiO2), the gene responsible for silicatein is strongly up- regulated, along with that for collagen.16 These experiments suggest that the sponge may be able to take advantage of silica concentrations as high as 60 mM, or alternatively, may be restricted in it’s growth, at levels below 60 mM.
It has been shown that in the sponge Halichondria panicea there is a correlation between the dissolved SiO2 in the seawater, and SiO2 content of the sponge.17 Moreover, it has been shown that silica uptake appears to require energy expenditure by the sponge, and that after conditions of starvation, uptake rates were greatly reduced. The ability of these sponges to remove SiO2 from the water column is sufficient so that in the summer they may actually compete with diatoms for available silicate.
In this same sponge, the rate of silica uptake is a function of the dissolved silica concentration in the seawater, with higher silica concentrations resulting in higher rates of uptake. The concentration at which the sponges take up silica at half of their maximal rate is 46 mM (2.8 ppm SiO2).14 Further, in certain waters it is believed that the growth of these sponges is limited by soluble silica, rather than by the availability of food. These sponges are capable of taking up 19 mmol/h per gram of tissue (maximally), so at 46 mM dissolved SiO2, they theoretically could take up 9.5 mmol/h per gram of tissue (though there is no evidence that the do take up silica that fast under normal growth conditions). A 50-gram sponge would then be able to take up 475 mmol/h, or 11.4 mmol (0.7 g) of silica in a day. A 100-gallon (378 L) aquarium with a high level of silica (30 mM or 1.9 ppm SiO2) only contains that much to begin with. Consequently, the potential
silica depletion in reef tanks with actively growing sponges could be substantial. Of course, if your sponges are not growing rapidly, then they likely are not using much silica. Likewise, if there is not much silica, then they may not be able to grow rapidly, even if other conditions are good.
This reason is, in fact, why I initiated dosing of silica (0.33 mM/day or 0.02 ppm SiO2/day) to my reef tank several years ago. I had a large sponge that I wanted to survive, and I had hoped that silica additions might alter the usual course of slow death for such sponges in most reef tanks. When I initiated that study, Julian Sprung told me that he had tried the same thing. In both of our cases the sponges eventually died, in my case lasting about 18 months. Perhaps we did not dose enough silica (0.33 mM/day is a small amount relative to the uptake numbers described above, and the concentration in the water column of my tank never rose above 0.8 mM (0.05 ppm SiO2) where it could be detected with a Hach kit). Alternatively, perhaps the food sources were not right or something else was wrong.
Marine Organisms that Use Silica: Mollusks
It turns out that the teeth (called radula) of many mollusks contain substantial silicon. The teeth are used for scraping algae from rocks (or glass in reef tanks). Consequently, they rapidly wear down and are quickly replaced. These radula are quite chemically complex, containing lots of different ions. The exact chemical composition is dependent on the species and family. The radula of limpets, such as Patella vulgata, contain large amounts of Si (up to 35%) and Fe (up to 51%), and substantial amounts of Al, Ca, K, Mg, Na, and P. The radula of chitons seem to be more variable, with Zn in particular varying substantially between species.18-21
It seems likely that these and other mollusks get the silicon necessary to form their radula from diatoms that they consume, rather than from the water column. However, if reef tanks are kept artificially low in silica, and do not have much in the way of diatoms for the mollusks to consume, it is a possibility that these organisms may become deficient in silicon. It has been claimed by some hobbyists that many mollusks do not live as long in reef aquaria as they do in the wild, although I’ve not seen any definitive data in this respect. Many hypotheses have been proposed, and it stands to reason that a deficiency in silicon might be considered as one of the possibilities, if, in fact, mollusks die prematurely in reef tanks.
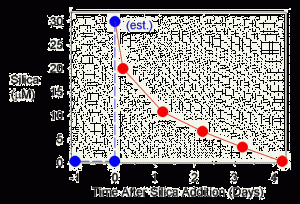
Figure 4. The concentration of soluble silica as a function of time after dosing. Dosing was done at t=0. The concentration for t=0 (30 mM) is an estimate based on what was added and the estimated total system volume.
Marine Organisms that Use Silica: Algae, Radiolarians, and Silicoflagellates
It has been reported that certain planktonic algae take up silica, but do not have an absolute growth requirement for it. One diatom specie, Phaeodactylum tricornutum (Bacillariophyceae) , and the prasinophyte Platymonas (a green flagellate), are two examples.22,23 These organisms have relatively inefficient silica uptake mechanisms, with half maximal uptake rates at 97 and 81 mM (5.9 ppm and 4.9 ppm SiO2) respectively. These values are much higher than for diatoms that require silica, which are typically 1-6 mM (0.06 – 0.4 ppm SiO2). Unlike most diatoms, Phaeodactylum tricornutum (Bacillariophyceae) seems to take up silica in the form of Si(OH)3O–, so it’s ability to take up silicate from a solution at fixed total silicic acid/silicate concentration is pH dependent. 24 What purpose the silica serves in these organisms isn’t known. Maybe it just makes the algae less palatable to herbivores.
Like diatoms, radiolarians are often beautiful organisms. They are protozoa that are nearly all planktonic. Most radiolarians have a siliceous skeleton, although some also contain strontium sulfate structures. 25 While radiolarians are a significant contributor to the silica cycle in the oceans, 26-28 I do not know how important they are in ordinary reef tanks. Other organisms also use silica, such as the silicoflagellates, but it is not evident to me whether they play any role in reef tanks either. 27
Measuring Silica
Before going on to discuss silica in reef tanks, a few comments on measurements of silica seem worthwhile. In the context of organisms that use silica, we are only interested in soluble forms of silica, typically silicic acid and silicate. Ignoring the fact that there can actually be other soluble forms in certain situations, like extended chains or rings, the most important distinction that reef keepers need to be aware of is between tests that analyze for silicon, regardless of form, and those that analyze for soluble silica.
Tests that analyze for silicon, such as ICP (inductively coupled plasma) can include silica particulates (e.g., fine sand) in the result. Even with filtration, fine particles can evade removal. Consequently, it is complicated to extrapolate from an ICP measurement to a soluble silicate concentration. In Ron Shimek’s tests of aquarium water,29 for example, the technique chosen was ICP. Consequently, people should not interpret the values obtained, 1.8 to 104 mM (0.05 to 2.9 ppm Si = 0.11 to 6.2 ppm SiO2 ) as necessarily indicating anything about the dissolved silica concentration present in the tanks studied (except that the dissolved silica cannot exceed those numbers).
Similarly, in studies of salt mixes,30 the different forms may be an issue as well. In that case, the authors attributed differences between ICP and wet chemistry methods to the nature of the silicon present.
Tests that analyze for soluble silica, such as any of the kits available to the hobby, will only detect soluble forms. I recommend the low range silica test from Hach, Model SI-7, catalog number 22550-00. While the values obtained with such kits may not be comparable to those obtained by other methods, they are suitable for understanding how much dissolved silica is present and available to organisms in tank water, and in other aqueous solutions, such as tap water.
Sources of Soluble Silica in Reef Tanks
What are the sources of soluble silica in reef tank? Certainly, tap water is a big one. It is added to many water supplies to raise the pH and reduce leaching of lead and copper into potable water from pipes.31 My tap water presently contains 17 mM (1.0 ppm SiO2). When I started my first reef tank years ago, that fact, and the fear of diatoms, was what drove me to set up an RO/DI system. In fact, many makers of RO/DI systems seem very concerned by silica, and they pass that concern along to their customers (and vice versa). Silica may, in fact, be too high in some water supplies. As it turns out, however, I presently do not believe that this silica level is too high to add to my tank. Rather, I think it would be beneficial. It turns out that when I intentionally dosed this amount (equivalent to a 2% daily evaporation rate) for 2 years, I did not experience any problems. It also amounts to less silica than I recommend people dose to their tanks at the end of this article.
[Nevertheless, I still use the RO/DI system to protect me from all of the many other compounds that could be present in the water that I don’t want in my tank.]

Figure 5. A view through the front glass of my reef tank 5 days after scraping the glass and adding the silica supplement
Other sources of soluble silica are much harder to pin down. Many foods and additives contain silicon in some form. Tests that have typically been used on foods, like ICP, cannot distinguish dissolved silica from tiny particulates, as mentioned above. In some of these tests, typical aquarium foods ranged from 20-540 ppm silicon. 32 A 5-gram spoonful of food that contained 540 ppm Si would actually only contain 2.7 mg of Si. If all of that were a soluble form, then putting it into a 100-gallon reef tank would raise the dissolved silica concentration by 0.25 mM (0.015 ppm SiO2). That value is small, but not insignificant. Still, without knowing whether it is soluble silica or not, one cannot tell whether it is a usable form. In any case, it is less than I recommend for dosing (see below).
Another potential source of silica is the artificial salt mix used to prepare the tank water. Despite the marketing hype in which many mixes claim to have no silica, most do have some (a few mM), and these levels are not unlike natural seawater.30 Nevertheless, that silica likely depletes in a few days in a real reef tank (see below).
Other sources may include any of the various supplements added to reef tanks. While there are far too many additives to consider, some that could be significant sources would include those that are added in the largest amounts to reef tanks: calcium and alkalinity supplements. Analyses by Craig Bingman33 and Greg Hiller34 of silica in calcium carbonate intended for CaCO3/CO2 reactors would suggest that little soluble silica is delivered in this fashion.
Limewater may also be a significant, but likely small source of soluble silica. According to the Mississippi Lime Company, their food grade lime35 contains 0.2% by weight silica, of which less than 0.1% is “crystalline silica”. If these values are accurate, and “non-crystalline silica” is actually a soluble form of some kind, then the addition of soluble silica to the tank can be calculated. A typical reef tank might add 1-2% of its volume in saturated limewater daily. Saturated limewater contains on the order of 1.5 g/L of Ca(OH)2, so it consequently contains about 3 mg/L of silica (assuming 0.2 wt percent SiO2 in the lime). At the daily delivery rate of 1-2% of the tank volume, the silica addition to the tank could be on the order of 0.5- 1 mM/day (0.03-0.06 ppm/day SiO2). That delivery is not insignificant relative to the few mM that
may be present in normal ocean water. I dose limewater in my tank (using Mississippi Lime Company quicklime, CaO, not the grade described here), and do not detect any soluble silica in my tank. Consequently, in my tank, this mechanism may be useful for adding soluble silica, but is apparently still small relative to the silica demand (see below).
One additional comment on limewater. At high pH, glass will etch as silica is dissolved into solution. From the standpoint of the glass involved, this does not become significant until the pH is significantly higher than that of limewater. But from the standpoint of small amounts of silica getting into the limewater, and then potentially into the tank, leaching from glass in the situation where limewater is dosed from a glass vessel may be significant.
The Dissolution of Quartz Sand
One of the issues that has been floating around the reef keeping hobby for a long time is the issue of whether “silica” sand actually releases soluble silica or not. It is remarkable that so many people have strong opinions on this issue, and yet so few people have ever bothered to do the easy experiment of measuring it. Many even fall for the trap of concluding that since their glass aquarium is not dissolving, then silica sand must not be either. All of the arguments against soluble silica being released from “silica” sand can be easily refuted, and I have done so in the past, but that is not the point of this article. Still, some background is worthwhile before getting to experimental results.

Figure 6. A view of the inside of the front glass of my reef tank 5 days after scraping the glass and adding the silica supplement.
Silica sand is largely composed of quartz. Quartz has a maximum solubility in pure freshwater of about 180 uM (11 ppm as SiO2)36, and is somewhat higher in seawater.37 That value is substantially in excess of the dissolved silica concentrations in any normal part of the ocean (excluding plumes from vents from hot springs and such). So why doesn’t quartz beach sand dissolve? It does, but it does so very slowly. The rate of dissolution of quartz has been studied, and it is very slow. 38 It is the slow dissolution of quartz, not the solubility itself, which allows it to remain on many ocean beaches.
A final comment on quartz sand is that it is known that organic acids can increase the rate of dissolution of quartz by at least a factor of ten.39 This may be especially applicable in reef tanks, where organic materials may be in abundance, particularly when organisms are living directly on the sand, potentially releasing such acids directly onto the sand surface.
The problem with extrapolating from the known very slow rate of dissolution of quartz to “silica sand” is that it simply is not pure quartz. The dissolution of soluble silica from “quartz sand” (98.5% SiO2) has long been known to exceed the solubility of quartz itself.40 Take a close look at some commercial “silica” sand. It isn’t even close to being white, which an absolutely pure quartz sand will be. There are all sorts of different colored particulates in it (some are even magnetic and can be picked out with a magnet). Without going into detail on mineralogy, suffice to say that there are many minerals that readily dissolve to release silicate into the water. Such dissolution is why freshwater rivers contain so much silica (typically 150 mM (9 ppm SiO2)).4 Your sand claims to be 98% quartz? What about that other 2%? Two percent of a 50-pound bag of sand is a pound of “other stuff”.
If you start with true beach sand, and don’t fracture it much, then it is very likely that you will detect little dissolution of silica from it in a few days (although I’ve not tried it), because most of the readily dissolved minerals would have disappeared long ago (or are trapped inside). But commercial play sands are not typically from beaches, and are not collected with any kind of gentleness. They are often mined from sand pits, crushed, screened, and generally treated rather roughly. This serves to break many of the grains, exposing new mineral inclusions that are then primed to dissolve. This source is, in my opinion, where most of the soluble silica comes from in “silica” sand.
So, on to some experiments. I bought some Quickcrete Play Sand from Home Depot and ran a number of tests on it. In all of the cases shown below the silica concentration was determined with a Hach low range silica kit after filtration through a 0.2 mm syringe filter. In cases where the concentration is above 1 ppm, the sample was diluted with RO/DI water prior to analysis. All experiments were carried out in the dark to reduce any effect due to diatom growth.

Figure 7. An organism (at arrow point) isolated from the glass surface after silica addition, tentatively identified as the diatom _Cylindrotheca closterium (Nitzschia closterium). It is the second most abundant in the collected material.
In the first experiment I took 3 cups of sand, and suspended it in 3 gallons of freshly made Instant Ocean salt mix that initially contained less than 0.8 mM of silica (0.05 ppm SiO2). After 48 hours of gentle stirring with a powerhead (the water was stirring, but not the sand), the silica concentration had risen to 17 mM (1.0 ppm SiO2).
I then rinsed the same sand 5 times with 1 gallon RO/DI water (1 minute each time), discarded the contents, and then ran the same stirring experiment with 2 new gallons of Instant Ocean salt mix. In 48 hours the silica concentration had again risen, this time to 15 mM (0.92 ppm SiO2). Then I let it sit unstirred for another 96 hours, and the concentration had risen more, to 23 mM (1.4 ppm SiO2).
In a different experiment, I took about 45 pounds of sand, and added 2 gallons of Instant Ocean salt mix. I let this mixture sit for 7 days, with once a day mixing with my hands for about 30 seconds. At then end of this test, the concentration was 90 mM (5.4 ppm SiO2).
It has been suggested that the amount of silica coming from calcerous sand might actually be as high or higher than that from silica sand. To test this hypothesis, I repeated the small-scale experiments above on a calcium carbonate sand from Home Depot (Southdown). In this case, there was some soluble silica released after the first 48 h, but only 1.6 mM (0.1 ppm SiO2), or about a factor of 10 lower than the silica sand. In a long-term test, the concentration had only risen to 5 mM (0.3 ppm SiO2) in 14 days with once a day stirring.
From these experiments, I conclude that:
- The “silica” play sand that I purchased from Home Depot can substantially raise the dissolved silica concentration in seawater.
- The dissolvable portion of the silica sand cannot be completely removed by several rinses with either fresh or salt water, although it may be decreased somewhat by that process.
- Southdown calcium carbonate sand (likely aragonite) can release soluble silica, but about ten fold less than the “silica” sand.
Is it OK to use silica sand? Probably. Many people do so. I also believe that not all “silica “ sands will be the same for the reasons described above relating to processing of the sand and the nature of the mineral inclusions present. So the fact that many people successfully use some (or many) types of silica sand does not necessarily imply that all people can use any type of “silica” sand without a problem.
In subsequent sections of this article I describe dosing recommendations for adding soluble silica. Is silica sand a good way to go from that perspective? I cannot really answer that. It probably provides some silica to reef tanks, but the amount is completely out of the control of the aquarist. For that reason alone, I believe that it would be a poor choice as the sole source of soluble silica for a reef tank. In a tank without any silica dosing, silica sand may, in fact, be more beneficial to the overall tank, at least from a silica delivery standpoint, than calcium carbonate sand. There are, of course, many other differences that might be the deciding factor on sand choice (color, texture, dissolution, particle size distribution, nutrient and metal binding properties of sands, etc). Many of these factors are more aesthetic than technical, and the technical ones are beyond the scope of this article.
The Effect of Added Soluble Silica on a Reef Tank
As I mentioned above, I added small amounts of sodium silicate to my tank for 2 years in order to see what effect it would have on a sponge that I was trying to maintain. The dosing was not successful in maintaining the sponge, but other facts learned from that experiment are instructive. Most importantly, I never noticed anything that I would call “diatoms.” The aquarium glass always turned green after a while, not shades of golden brown that are typically (but not always) associated with diatoms. Additionally, there was never any build up of detectable silica using the Hach test kit (which could readily detect 0.8 – 1.7 mM (0.05- 0.1 ppm SiO2).
When that bottle of sodium silicate solution ran out, I stopped dosing for a few months, with no obvious changes in the tank. Before going further, I should point out that my reef tank is fairly low in other nutrients. It has no nitrate detectable with LaMotte or Salifert kits, implying a concentration less than 8 mM (0.5 ppm). Inorganic phosphate detectable with the Hach kit was 0.3 mM (0.03 ppm). These figures are at the low end of values typically reported for reef tanks, but may be in excess of those present in surface ocean water.41 I also was routinely dosing Kent’s iron and manganese supplement during the entire course of the experiment, so iron limitation should not have been a factor.
Figure 1 shows the front glass of a portion of my 90-gallon aquarium a few hours after cleaning the glass with a razor blade. Figure 2 shows the same view 5 days after the cleaning. From this angle, the view is clearly somewhat obscured (I had actually taken photographs every day, but the 5 day point serves to show the differences after silicate was added). Figure 3 shows the inside of the glass, taken through the side of the tank. The growth is clearly green, and on again scraping the glass with a razor blade, the material that came off looked dark green

Figure 8. An organism (at arrow point) isolated from Tatu’s tank, tentatively identified as the diatom _Cylindrotheca closterium (Nitzschia closterium).
Then I added a spike of sodium silicate to the water, estimated to be about 30 mM (1.8 ppm SiO2; estimated because I knew the addition amount exactly, but not the actual water volume of my total system). Figure 4 shows the decline in the detectable silica concentration, again using the Hach kit, over the next 4 days. By day 4, the tank is back to where it started before the experiment, with none detectable (less than about 0.8 mM (0.05 ppm). Interestingly, the drop is quite rapid. The concentration drops by about half every day.
Figures 5 and 6 show the same views of the glass 5 days after dosing the silicate. In this case, the growth is clearly more yellow/brown that it was previously. However, the growth is no more obstructing of the view than when it was green algae that was growing. On scraping the glass with a razor blade, the growth appeared yellow brown as it came off.
I collected some of this material by filtration, and dried it on a filter paper (a process that severely damages the diatoms, but that was necessary to easily ship them overseas). I then sent them to Tatu Vaajalahti in Finland to take some photomicrographs and to see if they could be identified as diatoms. Identification of diatoms is a tricky business, especially for amateur diatom hunters like us. The range of possible structures in immense, and the ones that we found in greatest abundance did not obviously look like some of the pictures that we first located on line.
Figure 7 shows the second most abundant organism present. It bears a very close resemblance to the diatom _Cylindrotheca closterium (Nitzschia closterium).42 Tatu has found this in his tank water as well, and has taken better pictures of his own samples that have not been dried as mine were (Figure 8). In these photomicrographs, the color should be ignored as some of the samples were stained.

Figure 9. An assembly of organisms, possibly diatoms, isolated from the glass surface after silica addition. This organism is the most abundant in the collected material.
The most abundant organisms, shown in Figure 9, are also likely diatoms. They were identified by René van Wezel as being a small pennate diatom with platelike chloroplasts, possibly from genus Gomphonema. Together, these two organisms account for at least 80% of the total organisms, and large green algae ( > 100 mm) were few.
From these experiments, I conclude that:
- Silica can be a limiting factor for diatom growth in some reef tanks
- Adding soluble silica can increase diatom growth
- The increased diatom growth was not apparently in addition to, but in place of, green algae growth
- Added soluble silica is rapidly depleted from some reef tanks
- Taken together, these facts suggest that silica supplementation may be desirable
Silica Dosing Recommendations
Why would I recommend dosing silica? Largely because creatures in our tanks use it, the concentrations in our tanks (at least in mine) are below natural levels, and the sponges, mollusks, and diatoms may not be getting enough to thrive.
How much and what to dose?
I’d suggest dosing sodium silicate solution, as it is a readily soluble form of silica. It is very inexpensive. I initially used a high quality laboratory grade, but I’d expect the bulk grades sold to the world at large to be good enough (and I use it now). Remember, you aren’t dosing much, and the solutions available are very concentrated. You may find “water glass’ in certain stores because it is used by consumers for things like preserving eggs. Buying chemicals can be problematic for many people, however, and this hobby chemistry store 43 sells to individuals. Ten dollars (+ shipping) gets you enough to last 150 years of dosing with a 100-gallon tank, so cost is not an issue. I just ordered some from them myself and it came broken open, unfortunately. Some of you may have gotten Christmas presents that had ¾ of a gallon of sodium silicate solution coating them as they passed my package in the mail. Nevertheless, I
still have enough for several years!).
Many “water glass” or sodium silicate solutions are sold with the concentration indicated by “° Baume”. Degrees Baume is a measure of the specific gravity, and values in the 40’s are typical of these concentrated solutions.44 A concentration of 41° Baume equates to 29% SiO2 by weight. Note that the density is high (1.38 g/mL for 41° Baume), so volume measurements should take this into account. Maybe eventually, some of the hobby supplement manufacturers will provide a supplement.
Safety note: Sodium Silicate solution is very basic (high pH). In fact, the pH can be substantially higher than limewater, so it is very corrosive to tissue and to metal devices. Be careful to not spill it on yourself, wear some eye protection, and if you spill it on something metal, wash it. In all cases, extensive washing with water is recommended in case of spills or exposure.
Based on my dosing experience, aquarists are probably safe dosing the equivalent of 17 mM (1 ppm SiO2) once every 1-2 weeks. That is based on the fact that my tank used that much in less than 4 days without having any sort of “bad” reaction. Of course, there’s nothing wrong with starting at a tenth of that and ramping up. And, of course, if you do get too much in the way of diatoms, just back off on the dosing. I presume that all that I added to my tank went into various organisms that us it (sponges, diatoms, etc), but perhaps I have more sponges than other aquarists, and diatoms consequently may be more of a concern in some tanks than in mine.
I’d also advise occasionally checking the soluble silica concentration in the water, in case the demand in your tank is substantially less than mine. If the concentration started to rise above 50 mM (3 ppm SiO2), even in the absence of diatoms, I’d probably reduce the dose rate because that is close to the maximum concentration that surface seawater ever attains.
Here’s how to determine dosing amounts. I’ll assume that you want 17 mM (1 ppm SiO2) dosing, and you can scale from there. If the concentration of the supplement is 29% silica by weight (41° Baume), then it is 290,000 ppm silica. To get to 1 ppm silica, you then need to dilute by 290,000 fold. If you add 1.3 grams of this supplement (0.96 mL) to a tank with 100 gallons (378,500 mL), then the final concentration will be about 17 mM (1 ppm SiO2). I’d disperse the concentrated silicate solution into some fresh water before adding it to the tank, and then add it to a high flow area. Because the pH is high, you likely will see some cloudiness that is mostly magnesium hydroxide. The magnesium hydroxide will dissolve without a problem, but to be safe, add the supplement in a high flow area.
Happy Reefing!
References
- Silicon — Foe or Friend? By Craig Bingman http://www.animalnetwork.com/fish2/aqfm/2000/feb/features/1/default.asp
- A seasonal progression of Si limitation in the Pacific sector of the Southern Ocean. Nelson, David M.; Brzezinski, Mark A.; Sigmon, Daniel E.; Franck, Valerie M. College of Oceanic and Atmospheric Sciences, Oregon State University, Corvallis, OR, USA. Deep-Sea Research, Part II: Topical Studies in Oceanography (2001), 48(19-20), 3973-3995.
- Silicon limitation of biogenic silica production in the Equatorial Pacific. Leynaert, A.; Treguer, P.; Lancelot, Christiane; Rodier, Martine. Laboratoire “Flux de matiere et reponses du vivant”, Institut Universitaire Europeen de la Mer, Technopole Brest-Iroise, Plouzane, Fr. Deep-Sea Research, Part I: Oceanographic Research Papers (2000), Volume Date 2001, 48(3), 639-660.
- The silica balance in the world ocean: a reestimate. Treguer, Paul; Nelson, David M.; Van Bennekom, Aleido J.; DeMaster, David J.; Leynaert, Aude; Queguiner, Bernard. Unite Recherches Associee, Universite Bretagne Occidentale, Brest, Fr. Science (Washington, D. C.) (1995), 268(5209), 375-9.
- The silica budget in the sedimentary cycle. Siever, Raymond. Harvard Univ., Am. Mineralogist (1957), 42 821-41.
- Digital Eclipse Image Gallery: Diatom Frustules: http://www.microscopyu.com/galleries/dxm1200/diatomfrustulessmall.html, http://www.microscopyu.com/galleries/phasecontrast/surirellagemmasmall.html, http://www.nikon.co.jp/main/eng/news/2002/smallworld_e_02.htm
- Silicon availability and cell-cycle progression in marine diatoms. Brzezinski, Mark A.; Olson, Robert J.; Chisholm, Sallie W. Biol. Dep., Woods Hole Oceanogr. Inst., Woods Hole, MA, USA. Marine Ecology: Progress Series (1990), 67(1), 83-96.
- Silicon metabolism in diatoms: Implications for growth. Martin- Jezequel, Veronique; Hildebrand, Mark; Brzezinski, Mark A. Universite de Bretagne Occidentale, UMR 6539, CNRS, Institut Universitaire Europeen de la Mer, Technopole Brest-Iroise, Plouzane, Fr. Journal of Phycology (2000), 36(5), 821-840.
- Iron: A Look at Organisms Other than Macroalgae by Randy Holmes-Farley http://www.advancedaquarist.com/2002/10/chemistry
- The chemical form of dissolved Si taken up by marine diatoms. Del Amo, Yolanda; Brzezinski, Mark A. Department of Ecology, University of California at Santa Barbara, Santa Barbara, CA, USA. Journal of Phycology (1999), 35(6), 1162-1170.
- Modification of the biogeochemical cycle of silica with eutrophication. Conley, Daniel J.; Schelske, Claire L.; Stoermer, Eugene F. Cent. Environ. Estuarine Stud., Univ. Maryland Syst., Cambridge, MD, USA. Marine Ecology: Progress Series (1993), 101(1-2), 179-92.
- Chronic substrate limitation of silicic acid uptake rates in the western Sargasso Sea. Brzezinski, Mark A.; Nelson, David M. Marine Science Institute, University California, Santa Barbara, CA, USA. Deep-Sea Research, Part II: Topical Studies in Oceanography (1996), 43(2-3, Ocean Time-Series: Results from the Hawaii and Bermuda Research Programs), 437-453.
- Role of transient silicon limitation in the development of cyanobacteria blooms in the Guadiana estuary, south-western Iberia. Rocha, C.; Galvao, H.; Barbosa, A. CIMA-Centre for Marine and Environmental Research, FCMA- Faculdade de Ciencias do Mar e do Ambiente, Universidade do Algarve, Faro, Port. Marine Ecology: Progress Series (2002), 228 35-45.
- Silica uptake kinetics of Halichondria panicea in Kiel Bight. Reincke, T.; Barthel, D. Institut fur Meereskunde, Kiel, Germany. Marine Biology (Berlin) (1997), 129(4), 591-593.
- Learning from biological systems: novel routes to biomimetic synthesis of ordered silica structures. Cha, Jennifer N.; Shimizu, Katsuhiko; Zhou, Yan; Christiansen, Sean C.; Chmelka, Bradley F.; Deming, Timothy J.; Stucky, Galen D.; Morse, Daniel E. Dept. of Chemistry, University of CA, Santa Barbara, CA, USA. Materials Research Society Symposium Proceedings (2000), 599(Mineralization in Natural and Synthetic Biomaterials), 239-248.
- Expression of silicatein and collagen genes in the marine sponge Suberites domuncula is controlled by silicate and myotrophin. Krasko, Anatoli; Lorenz, Bernd; Batel, Renato; Schroder, Heinz C.; Muller, Isabel M.; Muller, Werner E. G. Institut fur Physiologische Chemie, Abteilung fur Angewandte Molekularbiologie, Universitat Mainz, Mainz, Germany. European Journal of Biochemistry (2000), 267(15), 4878-4887.
- Silica uptake of the marine sponge Halichondria panicea in Kiel Bight. Frohlich, H.; Barthel, D. Inst. Meereskunde, Kiel, Germany. Marine Biology (Berlin) (1997), 128(1), 115-125.
- Concentrations of elements in the radular teeth of limpets, chitons, and other marine mollusks. Okoshi, Kenji; Ishii, Toshiaki. Department of Biotechnology, Senshu University of Ishinomaki, Minamisakai, Japan. Journal of Marine Biotechnology (1996), 3(4), 252-7.
- Structure, morphology, composition and organization of biogenic minerals in limpet teeth. Mann, S.; Perry, C. C.; Webb, J.; Luke, B.; Williams, R. J. P. Sch. Chem., Univ. Bath, Bath, UK. Proceedings of the Royal Society of London, Series B: Biological Sciences (1986), 227(1247), 179-90, 6 plates.
- Mineralization and hardness of the radular teeth of the limpet Patella vulgata. Runham, N. W.; Thornton, Patrick R.; Shaw, David Anthony; Wayte, Richard C. Univ. Coll. North Wales, Bangor, Wales. Z. Zellforsch. Mikrosk. Anat. (1969), 99(4), 608-26.
- Morphology and mineral content of radula of chiton. Liu, Chuanlin; Zhao, Jiangao; Cui, Longbo; Liu, Xingjie. Department of Biochemistry, Yantai University, Yantai, Peop. Rep. China. Dongwu Xuebao (2001), 47(5), 553-557.
- Marine alga Platymonas species accumulates silicon without apparent requirement. Fuhrman, J. A.; Chisholm, S. W.; Guillard, R. R. L. Dep. Civ. Eng., Massachusetts Inst. Technol., Cambridge, Mass., USA. Nature (London) (1978), 272(5650), 244-6.
- Silicon uptake by algae with no known silicon requirement. I. True cellular uptake and pH-induced precipitation by Phaeodactylum tricornutum (Bacillariophyceae) and Platymonas sp. (Prasinophyceae). Nelson, David M.; Reidel, Gerhardt F.; Millan-Nunez, Roberto; Lara-Lara, J. Ruben. Coll. Oceanogr., Oregon State Univ., Corvallis, OR, USA. Journal of Phycology (1984), 20(1), 141-7.
- Silicon uptake by algae with no known silicon requirement. II. Strong pH dependence of uptake kinetic parameters in Phaeodactylum tricornutum (Bacillariophyceae). Riedel, Gerhardt F.; Nelson, David M. Coll. Oceanogr., Oregon State Univ., Corvallis, OR, USA. Journal of Phycology (1985), 21(1), 168-71.
- Biological minerals formed from strontium and barium sulfates. III. The morphology and crystallography of strontium sulfate crystals from the colonial radiolarian, Sphaerozoum punctatum. Hughes, N. P.; Perry, C. C.; Anderson, O. R.; Williams, R. J. P. Inorg. Chem. Lab., Univ. Oxford, Oxford, UK. Proceedings of the Royal Society of London, Series B: Biological Sciences (1989), 238(1292), 223-33, 3 plates.
- Vertical flux, ecology and dissolution of radiolaria in tropical oceans: implications for the silica cycle. Takahashi, Kozo. Woods Hole Oceanogr. Inst., Woods Hole, MA, USA. Avail. NTIS. Report (1981), (WHOI-81-103; Order No. PB82-199779), 465 pp. From: Gov. Rep. Announce. Index (U. S.) 1982, 82(17), 3410.
- Biological buffering of oceanic silica. Harriss, Robert C. Harvard Univ., Cambridge, Mass., USA. Nature (London) (1966), 212(5059), 275-6.
- Biosiliceous particle flux in the Southern Ocean. Abelmann, Andrea; Gersonde, Rainer. Alfred Wegener Inst. Pol. Mar. Res., Bremerhaven, Germany. Marine Chemistry (1991), 35(1-4), 503-36.
- It’s in the Water by Ron Shimek: http://www.reefkeeping.com/issues/2002-02/rs/feature/index.htm
- The Composition Of Several Synthetic Seawater Mixes by Marlin Atkinson and Craig Bingman: http://www.animalnetwork.com/fish2/aqfm/1999/mar/features/1/default.asp
- Lead in Drinking Water by EPA: http://www.epa.gov/ogwdw000/lead/passivation.htm
- Necessary Nutrition, Foods and Supplements, A Preliminary Investigation by Ron Shimek: http://www.animalnetwork.com/fish/data/foods.asp
- Calcium Carbonate for CaCO3/CO2 Reactors: More Than Meets the Eye by Craig Bingman: http://www.animalnetwork.com/fish2/aqfm/1997/aug/bio/default.asp
- Alternative Calcium Reactor Substrates by Greg Hiller: http://www.animalnetwork.com/fish/library/articleview2.asp?Section=Aquarium+Frontiers+–+Biochemistry+of+Aquaria&RecordNo=1571
- Vertical Calcium Hydroxide: CODEX Hydrated Lime: http://www.mississippilime.com/products/product.asp?dept%5Fid=310&pf%5Fid=31004&tabsetting=2
- Quartz solubility at low temperatures. Rimstidt, J. Donald. Dep. Geological Sciences, Virginia Polytechnic Inst. and State Univ., Blacksburg, VA, USA. Geochimica et Cosmochimica Acta (1997), 61(13), 2553-2558
- Quartz solubility in hydrothermal seawater: an experimental study and equation describing quartz solubility for up to 0.5 M NaCl solutions. Von Damm, K. L.; Bischoff, J. L.; Rosenbauer, R. J. Environ. Sci. Div., Oak Ridge Natl. Lab., Oak Ridge, TN, USA. American Journal of Science (1991), 291(10), 977-1007.
- The solubility of quartz. Van Lier, J. A.; De Bruyn, P L.; Overbeek, J. Th. G. Massachusetts Inst. of Technol., Cambridge, J. Phys. Chem. (1960), 64 1675-82.
- The dissolution of quartz in dilute aqueous solutions of organic acids at 25°C. Bennett, P. C.; Melcer, M. E.; Siegel, D. I.; Hassett, J. P. Dep. Geol., Syracuse Univ., Syracuse, NY, USA. Geochim. Cosmochim. Acta (1988), 52(6), 1521-30.
- The solubility of quartz and silicates. Lucas, C. C.; Dolan, M. E. Can. Med. Assoc. J. (1939), 40 126-34.
- The solubility of quartz and silicates. Lucas, C. C.; Dolan, M. E. Can. Med. Assoc. J. (1939), 40 126-34.
- Phosphorus: Algae’s Best Friend by Randy Holmes-Farley: http://www.advancedaquarist.com/2002/9/chemistry
- Cylindrotheca closterium (Nitzschia closterium) (Ehrenberg, 1841), http://www.dnr.state.md.us/bay/cblife/algae/diatom/cylindrotheca_closterium.h tml
- The Chemistry Store.com: http://chemistrystore.com/sodium_silicate.htm
- Sodium Silicate – Products and Specifications by PQ Corporation: http://www.pqcorp.com/productlines/SodiumSilicateSpecs.asp






0 Comments