Aluminum is an ion that doesn’t get much discussion in reefkeeping circles. It has little in the way of positive biological functions. I am not aware of any marine organisms with a demonstrated requirement for aluminum. It can, however, be toxic to marine organisms at elevated levels. Occasionally, folks discuss whether metal devices made from aluminum alloys will corrode in seawater. Most often, aluminum comes up during discussions of aluminum-based phosphate binding agents. These aluminum oxide materials have been reported to cause negative reactions in certain corals, and one hypothesis that has been suggested is that aluminum is released that irritates the corals.
In this article I will give some details of aluminum in natural seawater, and will discuss the sparse literature on the toxicity of aluminum ions to marine organisms. I will also show that aluminum is indeed released from one of these types of materials (Phosguard, sold by Seachem). Finally, I will show whether added soluble aluminum irritates any of several corals in a test aquarium.
Aluminum In The Ocean
Individual soluble aluminum ions in the ocean largely take the form Al(OH)4-, but some are also present as Al(OH)3.1,2 Aluminum is also strongly attracted to organics and some inorganics (like silica), making the exact speciation of aluminum very complicated. There is also a fair amount of aluminum present in particulate and colloidal forms (typically in combination with silica), ranging in concentration from about the same as the soluble fraction, to much greater.3-6
Interestingly, aluminum is present at much higher total concentration in the Gulf of Mexico (~0.002 ppm for particulate forms only),4 the Atlantic Ocean (0.00014 – 0.0016 ppm))8-12 and the Mediterranean Sea (0.00008 – 0.02 ppm)3 than in the Pacific Ocean (0.0000016 – 0.00016 ppm)7,8 or near Antarctica (0.00008 ppm).9 This difference provides a significant clue to the origin of most aluminum in surface seawater: airborne dust landing in the water.7,10-12 Dust from Africa is the proposed reason why the Atlantic is so much higher in concentration,9,12 while some in shore areas are also elevated due to the input from rivers.
The maximum solubility of aluminum at pH 8.2 in freshwater is about 2.7 ppm.13 That is, at concentrations higher than that, the aluminum will precipitate as amorphous aluminum hydroxide. I expect the solubility to be similar or higher in seawater, where complexation to organics may increase the solubility. Consequently, the solubility in both the oceans and in aquaria (as will be seen below) is apparently not typically limited by the solubility of aluminum hydroxide itself.
Biological Effects Of Aluminum: Toxicity
There are many known biological effects of aluminum, nearly all of which are negative.14 Aluminum toxicity has been extensively studied in fish, especially freshwater fish, but less so in other organisms, including marine fish.15 In freshwater systems, the toxicity of aluminum is a function of pH, with aluminum typically more toxic at lower pH. The reasons for this include the solubility, the speciation, and the nature of the interaction of aluminum with the surfaces of organisms as the pH changes.14 At pH 7, aluminum can bind to the gills of fish, inducing asphyxiation.15
Toxicity studies in marine systems has been much more limited. The table below describes some of the data:
| Species Tested | Endpoint | Concentration | Reference |
|---|---|---|---|
| Cancer anthonyi (a crab) | 7 day lethal concentration | 10 ppm | 16 and Web site |
| Crustaceans (4 species) | 3-4 day lethal concentration | 0.24-10 ppm | Web site |
| Mollusc | 3 day lethal concentration | 2.4 ppm | Web site |
| Ctenodrilus serratus (polychaetous annelid) | 4 day lethal concentration | 0.1 ppm | 17 and Web site |
| Capitella capitata (polychaetous annelid) | 4 day lethal concentration | 0.4 ppm | 17 and Web site |
| Neanthes arenaceodentata (polychaetous annelid) | 4 day lethal concentration | >0.4 ppm | 17 and Web site |
Biological Effects Of Aluminum: Uptake
In addition to suffering from overt toxicity, many organisms take up aluminum, and some have developed systems to deal with aluminum that they apparently don’t want. In freshwater snails, for example, it has been suggested that silica is used to detoxify aluminum:18,19
“These findings, and arguments on the stability, lability, and kinetics of aluminum-silicate interactions, suggest that a silicon-specific mechanism exists for the in vivo detoxification of aluminum,”19
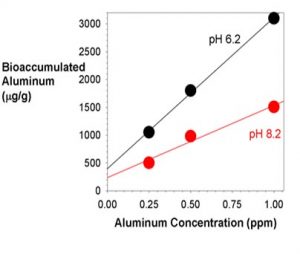
In marine systems, diatoms similarly take up aluminum and it is reported that it can impact their growth.20,21 The absorption of aluminum was recently studied in detail in the marine phytoplankton Dunaliella tertiolecta ( a unicellular green algae).22 The bioaccumulation was found to be strongly related to the aluminum concentration in the seawater (Figure 1). Consequently, one might expect that whatever problems aluminum causes, that it could be more severe as aluminum levels increase up to 1 ppm, at least for this particular organism. This result is important as that is the range of aluminum concentration that can result from exposure to Phosguard (later in this article).
Aluminum in Reef Aquaria
In a recent survey of 23 reef aquaria, Shimek claimed that aluminum levels ranged from 0.070 to 0.32 ppm, with a mean of 0.173 ppm .23 That same study claimed that Instant Ocean Contained 0.110 ppm aluminum. Other than values reported later in this article, I am not aware of any other published values for aluminum in reef aquaria.
These values were all generated by ICP (Inductively Coupled Plasma) where the sample is injected into a plasma and the light emissions of the various ions are quantified at one or more specific wavelengths unique to each element. I am skeptical that all of the values in the survey above represent real measurements of aluminum rather than noise in the ICP since they are all right around the limit of quantitation for aluminum in seawater. While this issue may seem esoteric, it is important to know how much aluminum is typically present in aquaria in order to understand whether the aluminum leached from products such as Phosguard represent a significant addition, or a trivial amount.
The following section discusses details of ICP testing for aluminum in seawater that may not interest most aquarists. It is intended to justify the skepticism about the measurements reported above. If one accepts the premise that those values may be unreliable, or if one just doesn’t care about such things, then skip on down to the next section, entitled “Inputs of Aluminum in Reef Aquaria: Salt Mixes.”
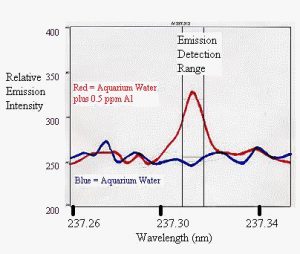
Figure 2: The ICP emission spectrum between 237.2 and 237.4 nm of a sample from my aquarium (blue) and the same sample after being spiked with 0.5 ppm aluminum (red).
As part of the experiments to test for the release of aluminum from Phosguard, I tested a sample of water from my aquarium as well as freshly made Instant Ocean (both at a salinity of 35 ppt). I used a new Varian ICP-OES in an attempt to detect aluminum in these samples (none were acidified or filtered unless otherwise noted). However, I was unable to detect anything but noise in these samples. That is, in looking at the emission spectra themselves, I simply saw a noisy background by eye, without any significant peak at the known emission wavelengths for aluminum (I used 237.312, 308.215, 394.401, and 396.152 nm). Spiked samples (standards made by adding soluble aluminum to the samples of aquarium water and Instant Ocean salt mix), and those where aquarium water or Instant Ocean water were exposed to Phosguard (below) did have clearly defined peaks at these wavelengths.
Figure 2 shows some typical ICP data for these samples. The blue line represents the emission spectrum displayed by my aquarium water sample. It is actually the average of three testing replicates taken from a single water sample. The red line represents the same sample with 0.5 ppm aluminum spiked into it. Clearly, the spiked aluminum gives a clearly defined signal exactly where it is supposed to be, while the aquarium water sample shows no such peak. This same conclusion applied at each wavelength examined, and for the Instant Ocean samples as well. From these data, I conclude that the concentrations of aluminum in my aquarium sample and in Instant Ocean are less than 0.05 ppm aluminum.
Nevertheless, the device software for the ICP is what most laboratories will use to quantify ions in high volume samples submitted for analysis. When I used the Varian software to determine the concentration in these samples, it integrated the noise in these samples to the equivalent of 0.05 – 0.12 ppm aluminum for both samples (and for each of several wavelengths used). Likewise, 18 mega-ohm deionized water (very pure) came out at 0.04 to 0.11 ppm aluminum. Consequently, the background noise, and the reality or not of the emissions being quantified must be carefully considered when reporting values near the noise limit for the device.
Was this an important issue for the samples analyzed for aluminum by Shimek? Is that why the samples all looked fairly similar in terms of aluminum concentration? Do any of those reported values represent real determinations, or simply background noise? I don’t know, but I am skeptical. Since I am not aware of any other measurements of aluminum in reef aquaria, I believe that we are left without knowing what the concentrations are, except that in the case of my aquarium at least, the concentration is ≤ 0.05 ppm.
Inputs of Aluminum in Reef Aquaria: Salt Mixes
In their study of artificial saltwater mixes, Atkinson and Bingman claimed that 8 different artificial salt mixes contained between 6 and 8 ppm aluminum (which they reported as 230-290 μmole/kg).24 Because the numbers are all so similar and so very much higher than my test (≤0.05 ppm for Instant Ocean) or those reported by Shimek (0.1 ppm for Instant Ocean), or the S-15 Report (0.006 ppm aluminum in Instant Ocean; similarly low for the other mixes tested), I suspect that the Atkinson and Bingman aluminum values may represent an artifact of some sort, either in testing or in data tabulation. Consequently, the starting artificial salt max may not be an especially important source of aluminum in aquaria (especially as compared to other inputs described below).
Inputs Of Aluminum In Reef Aquaria: Foods
Foods are, of course, another potential source of aluminum. In a study of the amounts of different elements in certain foods,25 Shimek presented the results shown in Table 2. The values have also been normalized to show the amount of aluminum in the foods in relation to the number of calories provided. Clearly, if aluminum is of primary concern, brine shrimp (highlighted in red) should probably not be on the menu.
If you fed 5 grams of it to a 100 gallon tank every day, that would amount to 5 g x 120 mg/kg = 0.6 mg/day or 219 mg/year. Added to that 100 gallons (379 L), that gives an addition of 219 mg/378 L/y = 0.6 ppm per year. The other listed foods would, of course, contribute much less. Unfortunately, these sorts of raw measures of aluminum say nothing about what form it is in. For example, it might be present as soluble aluminum, or as insoluble (particulate) forms.

Figure 4. The leather coral in the test aquarium after addition of aluminum. After the addition, it cycled between the look in this photograph, and that shown in Figure 3.
| Food | Calories/gram | Total Aluminum (ppm) | Aluminum (mg/kcalorie) |
|---|---|---|---|
| Formula One | 0.8 | 15 | 19 |
| Formula Two | 0.8 | 15 | 19 |
| Prime Reef | 0.8 | 11 | 14 |
| Lancefish | 0.9 | 9.8 | 11 |
| Brine Shrimp | 0.3 | 120 | 400 |
| Plankton | 0.7 | 8.1 | 12 |
| Gold Flakes | 4.2 | 80 | 19 |
| Tahitian Blend | 2.4 | 14 | 6 |
| Saltwater Staple | 3.6 | 95 | 26 |
| Nori | 3.6 | 83 | 23 |
| Golden Pearls | 3.9 | 49 | 13 |
Inputs Of Aluminum In Reef Aquaria: Calcium And Alkalinity Supplements
Another significant source of aluminum are the calcium and alkalinity supplements that aquarists use. In a recent paper on metals in aquaria,26 I quantified some of these inputs, and the results for aluminum are summarized below.
Limewater (kalkwasser) is made by dissolving calcium oxide or calcium hydroxide in water. The calcium oxide that I use from the Mississippi Lime Company is food grade, but still has certain impurities. The typical analysis of this material shows it to contain 0.10 % aluminum. It is not obvious what form this takes but since aluminum is quite soluble at pH 12.4 (total solubility = 80 ppm at pH 12.4,27 if saturated limewater were made from CaO with 0.1% aluminum, it would contain 1 ppm aluminum) it is a reasonable hypothesis that it dissolves into the limewater and is delivered to the aquarium. If one were adding 2% of the aquarium volume in saturated limewater (0.0204 moles/L CaO) every day for a year, one would have added the equivalent of 8.3 ppm aluminum.
The amounts of metals that are added to an aquarium when using a CaCO3/CO2 reactor can also be determined. The impurities present in such media varies with the source or brand of the media, as has been shown in different articles by Craig Bingman 28 and Greg Hiller 29. If we make the assumption that we want the same total amount of calcium and alkalinity as in the limewater case described above, then we can calculate the following amounts of metals added over a year:
| Substrate | Amount added in 1 year (ppm) |
|---|---|
| Conklin Limestone | <0.001 |
| Nature’s Ocean | 1.2 |
| Koralith | 1.0 |
| Super Calc Gold | 1.1 |
As can be seen, the amount added over the course of a year can be quite substantial, but is less than is delivered via limewater (at least in these calculations). To be honest, I’m not sure why there is less aluminum delivered by CaCO3/CO2 reactors than limewater. Calcium oxide is made by heating calcium carbonate until the carbon dioxide is driven off. Such a conversion shouldn’t impact aluminum concentrations. Perhaps the difference simply reflects artifacts in one or both of the testing methods, contaminants in production, or in the nature of the original calcium carbonate chosen for each process.
Other methods of calcium and alkalinity addition presumably also deliver some amount of aluminum, though I’ve not seen any analysis of any of them to comment further.
Inputs of Aluminum in Reef Aquaria: Phosguard
Many aquarists claim to see undesirable effects on corals when using Phosguard, made by Seachem. Many aquarists have attributed that effect to released aluminum, since it is largely composed of aluminum oxide (possibly with silicon present too). In the first phase of testing that hypothesis, I examined whether Phosguard does indeed release any aluminum into solution.
Table 4 summarizes the results for a serious of samples in which commercial Phosguard (75 mL) was placed into contact with aquarium water or freshly made Instant Ocean artificial seawater (500 mL). The samples we allowed to sit in closed plastic containers. Once every 3 days or so the containers where gently shaken for a few seconds. Aliquots were removed, in some cases filtered through a 0.45 μm filter to remove “particulates”, and the aluminum was determined by ICP (without acidification). The concentrations were determined by comparison to standard made by spiking 0.5 ppm aluminum into aquarium water or Instant Ocean artificial seawater (which had been shown earlier in this article to have no detectable aluminum). All of the samples had a clearly definable emission peak in the appropriate place, although the lowest sample (0.06 ppm) is close to the limit of detection.
| Water Sample | Exposure Time | Filtration | Aluminum Concentration (ppm) |
|---|---|---|---|
| Aquarium Water | none | none | ≤ 0.05 |
| Aquarium Water | 1 week | none | 0.37 |
| Aquarium Water | 1 week | 0.45 μm | 0.06 |
| Aquarium Water | 5 weeks | none | 0.71 |
| Aquarium Water | 5 weeks | 0.45 μm | 0.12 |
| Instant Ocean | none | none | ≤ 0.05 |
| Instant Ocean | 1 week | none | 1.11 |
| Instant Ocean | 1 week | 0.45 μm | 0.13 |
From the results in Table 4 it is evident that Phosguard does release aluminum to the water, and that the majority of this is present in particulate form (that is, that it is removed on a 0.45 μm filter (although that does not demonstrate that it was originally released as particulates).
In order to determine if these results are caused primarily by fine particles that come with the much larger Phosguard particles (typically about 2 mm spheres), a batch was rinsed very thoroughly with RO/DI water (8 times, with each rinse lasting about 1 minute and each rinse volume comprising about 20 times the solid particle volume). These rinsed Phosguard particles were then exposed to aquarium water as above. The results are shown in Table 5.
| Water Sample | Exposure Time | Filtration | Aluminum Concentration (ppm) |
|---|---|---|---|
| Aquarium Water | none | none | ≤ 0.05 |
| Aquarium Water | 2 weeks | none | 0.25 |
| Aquarium Water | 2 weeks | 0.45 μm | 0.16 |
Not surprisingly, the concentration is reduced in the unfiltered sample, indicating that the rinsing may well have removed some fine particles that were contributing to the results in the unfiltered samples. However, the aluminum concentration in the filtered sample is not reduced, indicating that the “dissolved” fraction of the aluminum is not altered by rinsing the Phosguard first.
Significance Of Aluminum Release From Phosguard
Is the amount of aluminum released from Phosguard significant? Moreover, is it adequate to explain the results on corals that have been reported by aquarists? This question is extremely difficult to answer without some biological experiments. The tests run above show reasonably high concentrations of aluminum. Possibly high enough to cause problems for the organisms shown in Table 1 . But these tests were carried out on a large amount of Phosguard in a small amount of water. Tests with larger volumes of water might well result in lower aluminum concentrations. Additionally, the exact nature of the aluminum in these tests may well be different than in the toxicity tests reported above. That is, the nature may be particulate vs. colloidal vs. soluble vs. complexed by organics, etc.
Biological Testing Of Aluminum Exposure
In order to more definitively show whether aluminum released by Phosguard might be the cause of reaction in corals, it seems prudent to test aluminum on corals. Toward this end, I set up a 30-gallon aquarium with several corals. These included a leather coral ( Sarcophyton sp.), green star polyps, and brown mushroom corals. In addition, the tank contained sand, live rock, and some macroalgae ( Chaetomorpha sp. and Caulerpa racemosa ).
Pictures were taken of the several corals, and then aluminum was added. Figure 3 shows the leather coral before any addition. The first aluminum addition boosted the aluminum concentration by 0.005 ppm by adding 0.15 mL of an aluminum chloride solution to the water. A pH electrode was in the water at the time and recorded no pH change (pH = 8.32). There was no apparent change in any corals in 2 hours.
The second aluminum addition boosted the aluminum concentration by 0.045 ppm by adding 1.35 mL of an aluminum chloride solution to the water (total additions = 0.05 ppm aluminum). A pH electrode was in the water at the time and recorded a pH drop of from 8.35 to 8.33-8.34 pH. There was no apparent change in any corals in 24 hours.
The third and last aluminum addition boosted the aluminum concentration by 0.45 ppm by adding 1.35 mL of an aluminum chloride solution to the water (total additions = 0.5 ppm aluminum). A pH electrode was in the water at the time and recorded a pH drop of from 8.35 to 8.25 pH. There was no apparent change in any corals in 1 hour. By the 5-hour mark, the leather coral had closed (pH = 8.30), and then it began cycling between open and closed every hour or two, continuing into the next day. Figure 4 shows the closed form.
Thirty six hours after the last aluminum addition, the leather was still cycling between open and closed. This behavior had not been exhibited by this coral prior to aluminum additions. While I cannot be certain it was a result of the treatment, it seems likely.
After 48 hours, the leather no longer opened at all. It then stayed closed for the next 3 days until the termination of the experiment. I’ve since moved it to my main tank in the hope that it will recover.
5 hours after the last aluminum addition, the mushroom corals appeared less expanded than before the aluminum additions, but not nearly as dramatically as the leather. They stayed that way until the termination of the experiment.
The green star polyps seemed unchanged for the first 48 hours. After that, they expanded significantly less than they had previously. The polyps were about half of the size that they were before dosing aluminum. They were still that way at the termination of the experiment.
Conclusions
Aluminum is an ion that does not get much attention, and has no clear biological use in aquaria. It can, however, have an impact on aquarium organisms if elevated sufficiently over natural levels. Phosguard has been shown to release aluminum to artificial seawater. Further, it appears that the release of aluminum could be the cause of the effects that some folks have seen in aquaria when using aluminum-based phosphate and silicate absorbing materials. However, only a larger study could definitively demonstrate that to be the case.
Such biological effects have not been widely reported for the iron-based phosphate removers (e.g., Rowaphos and Salifert’s Phosphate Killer). Consequently, if you are interested in using phosphate-absorbing media, those latter types might be a better choice.
Happy Reefing!
References
- Chemical Oceanography, Second Edition. Millero, Frank J.; Editor. USA. (1996), 496 pp. Publisher: (CRC, Boca Raton, Fla.)
- Effects of salinity, sulfate and carbonate on the solubility and speciation of aluminum, barium and copper in seawater. Sadiq, Muhammad. Res. Inst., King Fahd Univ. Pet. Miner., Dhahran, Saudi Arabia. Environmental Technology Letters (1988), 9(9), 1021-8.
- Aluminum in the south-eastern Mediterranean waters off the Egyptian coast. El-Nady, F. E.; Dowidar, N. M. Oceanography Department, Faculty of Science, Alexandria University, Alexandria, Egypt. Estuarine, Coastal and Shelf Science (1997), 45(3), 345-355.
- Distribution of particulate aluminum in the Gulf of Mexico. Feely, R. A.; Sackett, W. M.; Harris, J. E. Dep. Oceanogr., Texas A and M Univ., College Station, TX, USA. Journal of Geophysical Research (1971), 76(24), 5893-902.
- Colloidal aluminum and iron in seawater: An intercomparison between various cross-flow ultrafiltration systems. Reitmeyer, Rebecca; Powell, Rodney T.; Landing, William M.; Measures, Christopher I. Department of Oceanography, University of Hawaii at Manoa, 1000 Pope Road, Honolulu, HI 96822, USA. Marine Chemistry (1996), 55(1/2), 75-91.
- Dissolved-particulate interactions of aluminum in ocean waters. Moore, R. M.; Millward, G. E. Dep. Oceanogr., Dalhousie Univ., Halifax, NS, Can. Geochimica et Cosmochimica Acta (1984), 48(2), 235-41.
- The biogeochemistry of aluminum in the Pacific Ocean. Orians, Kristin J.; Bruland, Kenneth W. Inst. Mar. Sci., Univ. California, Santa Cruz, CA, USA. Earth and Planetary Science Letters (1986), 78(4), 397-410.
- Dissolved aluminum in the Weddell Sea. Moran, S. B.; Moore, R. M.; Westerlund, S. Dep. Oceanogr., Dalhousie Univ., Halifax, NS, Can. Deep-Sea Research, Part A: Oceanographic Research Papers (1992), 39(3-4A), 537-47
- Aluminum in the South Atlantic: Steady state distribution of a short residence time element. Measures, C. I.; Edmond, J. M. Dep. Earth, Atmos. Planet. Sci., Massachusetts Inst. Technol., Cambridge, MA, USA. Journal of Geophysical Research, [Oceans] (1990), 95(C4), 5331-40.
- Dissolved aluminum in the central North Pacific. Orians, Kristin J.; Bruland, Kenneth W. Cent. Mar. Stud., Univ. California, Santa Cruz, CA, USA. Nature (London, United Kingdom) (1985), 316(6027), 427-9.
- Distribution of beryllium, aluminum, selenium, and bismuth in the surface waters of the western North Atlantic and Caribbean. Measures, C. I.; Grant, B.; Khadem, M.; Lee, D. S.; Edmond, J. M. Dep. Earth, At. Planet. Sci., Massachusetts Inst. Technol., Cambridge, MA, USA. Earth and Planetary Science Letters (1984), 71(1), 1-12.
- The role of dust deposition in determining surface water distributions of Al and Fe in the South West Atlantic. Vink, S.; Measures, C. I. Department of Oceanography, University of Hawaii, Honolulu, HI, USA. Deep-Sea Research, Part II: Topical Studies in Oceanography (2001), 48(13), 2787-2809.
- Aquatic Chemistry Concepts. Pankow, J. F. (1991), 712 pp. Publisher: Lewis Publishers, Inc.
- Environmental hazards of aluminum to plants, invertebrates, fish, and wildlife. Sparling, Donald W.; Lowe, T. Peter. National Biological Service, Patuxent Environmental Science Center, Laurel, MD, USA. Reviews of Environmental Contamination and Toxicology (1996), 145 1-127.
- Ecotoxicology of aluminum to fish and wildlife. Sparling, Donald W.; Lowe, T. Peter; Campbell, Peter G. C. U.S. Geological Survey, Patuxent Wildlife Research Center, Laurel, MD, USA. Editor(s): Yokel, Robert A.; Golub, Mari S. Research Issues in Aluminum Toxicity (1997), 47-68.
- Acute toxicities of eleven metals to early life-history stages of the yellow crab Cancer anthonyi. Macdonald, J. M.; Shields, J. D.; Zimmer-Faust, R. K. Mar. Sci. Inst., Univ. California, Santa Barbara, CA, USA. Marine Biology (Berlin, Germany) (1988), 98(2), 201-7.
- Effects of aluminium and nickel on survival and reproduction in polychaetous annelids. Petrich, Stephen M.; Reish, Donald J. Dep. Biol., California State Univ., Long Beach, CA, USA. Bulletin of Environmental Contamination and Toxicology (1979), 23(4-5), 698-702.
- Influence of oligomeric silicic and humic acids on aluminum accumulation in a freshwater grazing invertebrate. Desouky, M. M.; Powell, J. J.; Jugdaohsingh, R.; White, K. N.; McCrohan, C. R. School of Biological Sciences, University of Manchester, Manchester, UK. Ecotoxicology and Environmental Safety (2002), 53(3), 382-387.
- Aluminum-dependent regulation of intracellular silicon in the aquatic invertebrate Lymnaea stagnalis. Desouky, Mahmoud; Jugdaohsingh, Ravin; McCrohan, Catherine R.; White, Keith N.; Powell, Jonathan J. School of Biological Sciences, University of Manchester, Manchester, UK. Proceedings of the National Academy of Sciences of the United States of America (2002), 99(6), 3394-3399.
- Biological control of dissolved aluminum in seawater: experimental evidence. Stoffyn, Marc. Dep. Geol. Sci., Northwestern Univ., Evanston, IL, USA. Science (Washington, DC, United States) (1979), 203(4381), 651-3.
- Kinetics of the removal of dissolved aluminum by diatoms in seawater: A comparison with thorium. Moran, S. B.; Moore, R. M. Dep. Mar. Chem. Geochem., Woods Hole Oceanogr. Inst., Woods Hole, MA, USA. Geochimica et Cosmochimica Acta (1992), 56(9), 3365-74.
- Bioaccumulation of aluminium in Dunaliella tertiolecta in natural seawater: aluminium-metal (Cu, Pb, Se) interactions and influence of pH. Sacan, M. Turker; Balcioglu, I. Akmehmet. Department of Biology, Istanbul University Faculty of Science, Istanbul, Turk. Bulletin of Environmental Contamination and Toxicology (2001), 66(2), 214-221.
- It’s (in) the Water by Ronald L. Shimek. Reefkeeping.com. Volume 1. Number 1. February 2002 http://reefkeeping.com/issues/2002-02/rs/feature/index.htm
- The Composition Of Several Synthetic Seawater Mixes by Marlin Atkinson and Craig Bingman Aquarium Frontiers March 1999. http://www.animalnetwork.com/fish2/aqfm/1999/mar/features/1/default.asp
- Necessary Nutrition, Foods and Supplements, A Preliminary Investigation by Ronald L. Shimek. Aquarium Fish Magazine. 13: 42-53.
- Reef Aquaria with Low Soluble Metals. By Randy Holmes-Farley, Reefkeeping, April 2003 http://reefkeeping.com/issues/2003-04/rhf/feature/index.htm
- Metals In Limewater by Randy Holmes-Farley, Advanced Aquarist, May 2003 http://www.advancedaquarist.com/2003/5/chemistry
- Calcium Carbonate for CaCO3/CO2 Reactors: More Than Meets the Eye by Craig Bingman Aquarium Frontiers, August 1997.
- Alternative Calcium Reactor Substrates by Greg Hiller Aquarium Frontiers.




0 Comments